The first disease biomarkers discovered in saliva cells
For years, the challenge in saliva diagnostics has been the low and highly variable biomarker concentration in the standard specimen — the saliva fluid.
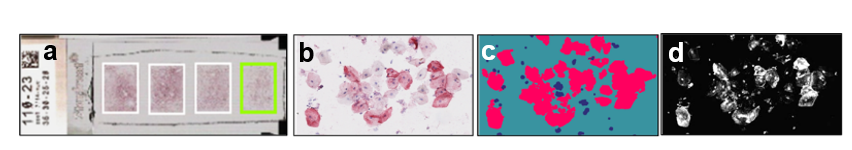
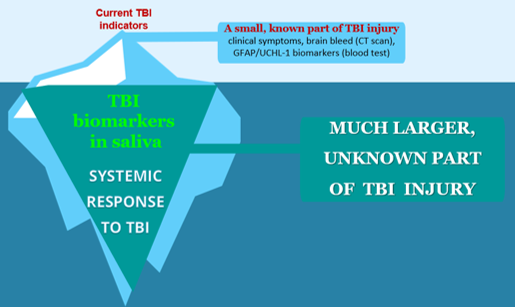
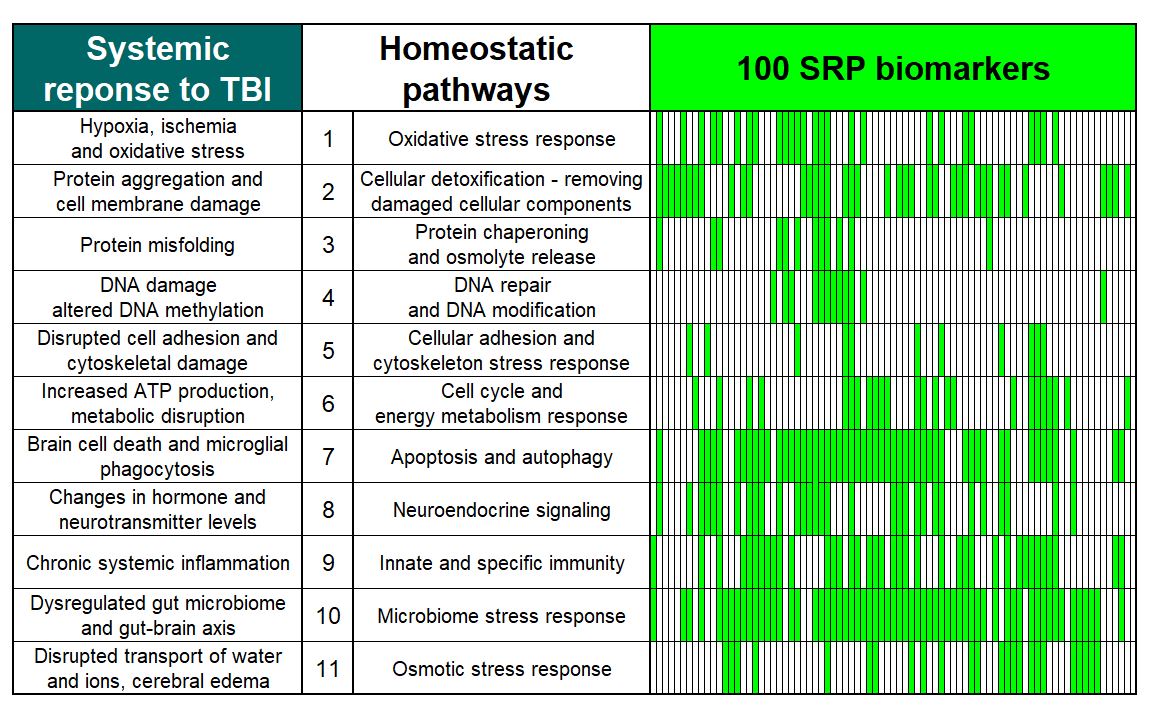
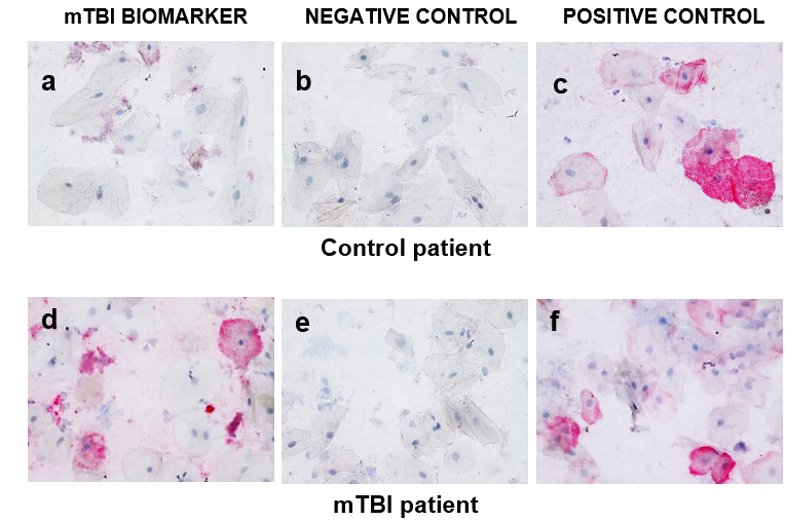
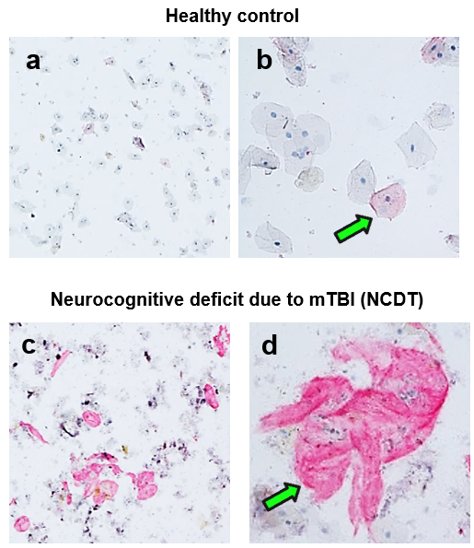
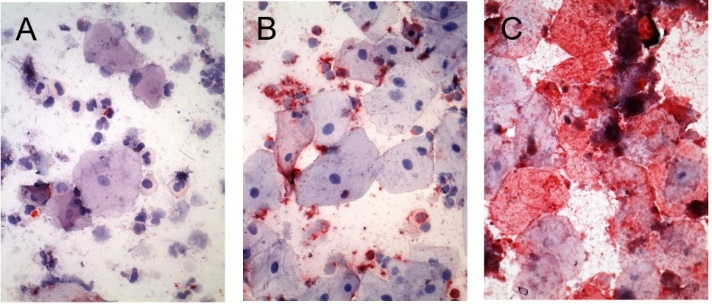
We have fundamentally changed saliva diagnostics by showing that Saliva contains a high concentration of live cells expressing disease biomarkers.
ADVANTAGE: Saliva with cells provides >100 times higher and 7-fold less variable biomarker concentration than saliva fluid.
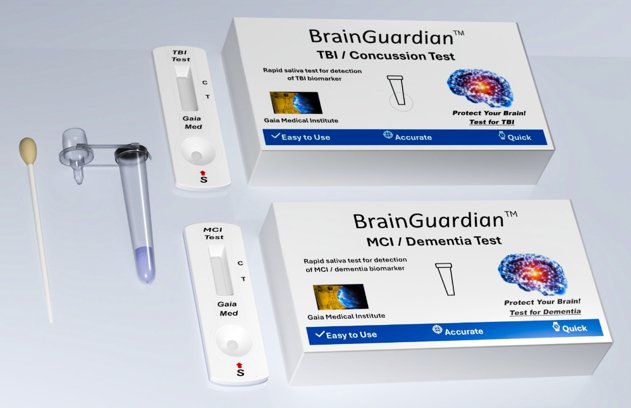
This breakthrough allows our BrainGuardian™ to detect the saliva-based TBI biomarker using a rapid lateral flow test — no laboratory equipment required.
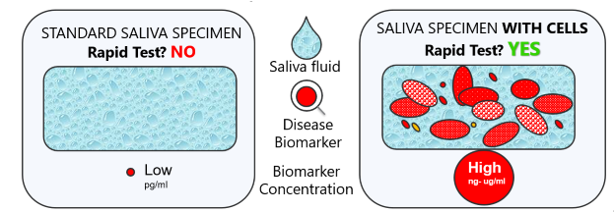
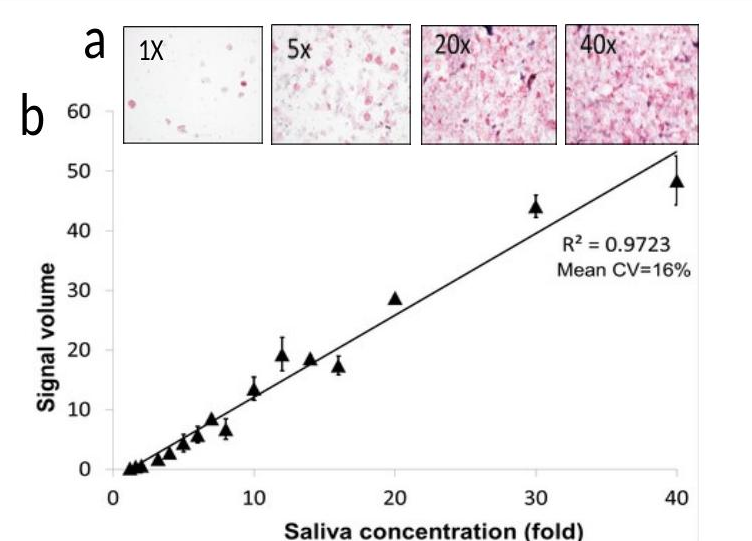
Standard saliva fluid yields low biomarker concentration (pg/ml). Cellular saliva yields high concentration (ng–μg/ml), enabling a rapid visual test.
Original magnification ×100
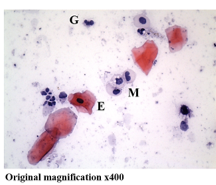
Original magnification ×400
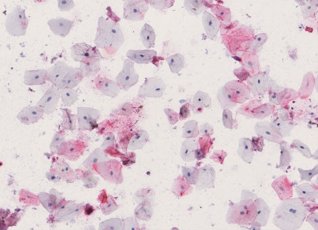
Cellular composition of human saliva. Hematoxylin & Eosin stain. Adult human saliva contains approximately 2 million cells/ml comprising epithelial cells (E), granulocytes (G), monocytes (M) and lymphocytes.
References
S. Southern, J Hum Virol. (1998) 1(5):328-37. Milk-borne transmission of HIV. Characterization of productively infected cells in breast milk and interactions between milk and saliva.
Southern, S. et al. (2002). Cellular Mechanism for Milk-Borne Transmission of HIV and HTLV. In: Advances in Experimental Medicine and Biology, vol 503. Springer, Boston, MA. https://doi.org/10.1007/978-1-4615-0559-4_21